Immunology
Abstract
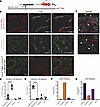
Brain metastases (BrMs) occur in approximately 30% of cancer patients, causing nearly one-fifth of cancer deaths. While immune checkpoint inhibitors (ICIs) benefit some BrM patients, responses remain highly variable. This variability partly reflects distinct histopathological growth patterns that include minimally invasive (MI) and highly invasive (HI) brain BrMs. Here we show that MI BrMs exhibit robust immune infiltration, whereas HI lesions are immunosuppressed. However, histological differentiation between MI and HI can be challenging because of subjective margin assessment. Here, using highly multiplexed spatial proteomics on 119 tumor sections from 46 patients with BrMs, we identify CHI3L1 as a key mediator of the immunosuppressive microenvironment in HI BrMs. In preclinical models, genetic deletion of CHI3L1 converts immune-cold metastases into lymphocyte-rich, ICI-responsive lesions infiltrated by granzyme B+ CD8+ T cells. In BrM patients treated with ICI, immunohistochemical quantification of CHI3L1 expression was a stronger predictor of ICI response than traditional MI/HI classification. Thus, CHI3L1 represents a promising biomarker and therapeutic target for BrMs.
Authors
Sarah M. Maritan, Elham Karimi, Matthew Dankner, Aldo Hernandez-Corchado, Miranda W. Yu, Matthew G. Annis, Yashar Aghazadeh Habashi, Morteza Rezanejad, Bridget Liu, Nebras Koudieh, Emilie Pichette, Parvaneh Fallah, Benoit Fiset, Yuhong Wei, Ali Nehme, Chun Geun Lee, Jack A. Elias, Morag Park, Yasser Riazalhosseini, Hamed Najafabadi, Kevin Petrecca, Marie-Christine Guiot, Daniela F. Quail, Logan A. Walsh, Peter M. Siegel
Abstract
Bladder cancer (BCa) mortality is mainly driven by metastatic dissemination and an immunosuppressive tumor microenvironment. Here, we identify ELN (tropoelastin), an extracellular matrix protein abundantly secreted by cancer-associated fibroblasts (CAFs), as a critical determinant of these processes and a marker of poor prognosis. ELN promotes epithelial-mesenchymal transition (EMT), facilitates lymphatic spread, and induces immune dysfunction characterized by macrophage polarization toward an M2 phenotype and T cell exhaustion. Mechanistically, ELN functions as a binding partner of TGF-β receptor 2 (TGFBR2), thereby triggering SMAD2/3-dependent TGF-β1 secretion and establishing a feed forward signaling loop. This ELN/TGFBR2/TGF-β1 axis amplifies metastatic capacity and immunosuppressive signaling, ultimately accelerating disease progression and diminishing responsiveness to immune checkpoint blockade. Functional studies in BCa organoids and murine models demonstrated that pharmacologic blockade of the ELN-TGFBR2 interaction effectively suppressed tumor metastasis and restored antitumor immunity. Collectively, our findings establish ELN as a CAF-derived driver of metastasis and immune evasion in BCa. Targeting the ELN-TGFBR2 interaction offers a promising therapeutic strategy to limit metastatic progression and enhance the efficacy of immunotherapy in this lethal disease.
Authors
Wentao Xu, Jia Gao, Shanshan Wu, Jianshang Huang, Chenchen An, Chonggui Jiang, Nianping Liu, Chen Cheng, Zihan Wang, Zijian Dong, Yuchen Xu, Jun Zhou, Hanren Dai, Xiaolei Li, Honghai Xu, Songyun Zhao, Qianwen Fan, Yang Li, Ying Dai, Li Zuo, Hua Wang
Abstract
HLA-E-restricted HIV-specific T cells offer exciting possibilities for immunotherapy. However, HLA-E binding peptides are rare. A recent study showed that in HLA-B*57:01 people living with HIV (PLWH), the peptide that dominates the T cell response, KAFSPEVIPMF (KF11), also stimulates HLA-E-restricted T cells, even though direct binding of this peptide to HLA-E could not be demonstrated. We therefore changed position 2 alanine for methionine in the peptide (referred to as KMF11) which greatly enhanced binding to HLA-E. This enabled the generation of stabilised HLA-E-KMF11 tetramers which were used to select and then grow specific T cell clones from T cells of HLA-B*57:01 negative blood donors primed with this peptide in vitro. Approximately 20% of these T cell clones reacted with HLA-E positive cells presenting the native KF11 peptide. Furthermore, these T cells inhibited replication of HIV-1 NL4-3 in CD4 T cells in vitro. Therefore, this native peptide can be presented by HLA-E to CD8 T cells, although priming in vivo may depend on cross reactivities to classical MHC Ia types. Nevertheless, such T cells could be exploitable for immunotherapy given the conservation of this HIV1 peptide epitope and the non-polymorphism in HLA-E.
Authors
Hong Sun, Hongbing Yang, Max N. Quastel, Simon Brackenridge, Wanlin He, Anna E. Kliszczak, Margarida Rei, Persephone Borrow, Geraldine M. Gillespie, Andrew J. McMichael
Abstract
Kawasaki disease (KD) is an acute febrile systemic vasculitis of unknown etiology and the leading cause of acquired heart disease among children. Complement activation has long been observed in patients with acute KD, however, its contribution to disease development remains unknown. Here, using publicly available datasets, we showed that patients with acute KD exhibited higher expression of complement products in whole blood, consistent with the activation of the complement pathway. Similarly, in the Lactobacillus casei cell wall extract (LCWE) murine model of KD, LCWE injection induced increased expression of complement products in cardiovascular tissues, suggestive of activation of the complement pathways. C3-deficient mice or WT mice treated with the complement C5a Receptor 1 (C5ar1) antagonist developed significantly more severe LCWE-induced cardiovascular lesions and vasculitis. Furthermore, we observed that LCWE binds to serum C3, an opsonizing factor that labels microbial targets for clearance, and LCWE deposition in the liver was significantly higher in C3-deficient mice compared to WT mice. Overall, our data indicate that blocking the complement system significantly exacerbates LCWE-induced KD vasculitis, likely by impairing C3-mediated clearance of LCWE. These data suggest that the complement pathway may play a protective role in KD pathogenesis by promoting clearance of potential bacterial or viral trigger of KD.
Authors
Asli E. Atici, Begüm Kocatürk, Benjamin L. Ross, Emily A. Aubuchon, Rebecca A. Porritt, Thacyana T. Carvalho, Takahiro Namba, Youngho Lee, Magali Noval Rivas, Moshe Arditi
Abstract
Antibody production by B cells has emerged as an important factor in regulating anti-tumor immunity with both suppressive and promotive roles in cancer. However, the specific impact of antibody deficiency during development of pancreatic ductal adenocarcinoma (PDAC) has not been explored. To address this question, we crossed the well-established KPC mouse model to mice lacking all circulating immunoglobulin (Ig) due to genetic ablation of both Ig secretion and Ig class switching (KPC-μSAID mice). KPC-μSAID mice exhibited a two-fold acceleration in tumor formation, a two-fold reduction in median survival, and increased liver metastases versus KPC-WT control mice. Immunofluorescence analysis of pancreatic tissues from antibody-sufficient KC- and KPC-WT mice showed that IgG was predominantly localized within extracellular matrix (ECM). Furthermore, in both KC- and KPC-μSAID mice, ECM density and podoplanin+ cancer-associated fibroblasts (CAFs) were significantly reduced. In the KPC-μSAID tumor microenvironment (TME), intratumoral myeloid-derived suppressor cells (MDSC) were also increased, while CD4+ and CD8+ T cells decreased, relative to tumor-bearing KPC-WT mice, with macrophage exhibiting a mixed polarization phenotype. These findings were recapitulated in antibody-subclass-deficient, KPC-AID mice, suggesting a potentially novel function of IgG in suppressing PDAC progression, by directly or indirectly regulating pancreatic fibrosis and the density of the ECM.
Authors
Jeremy B. Foote, Sujith Sarvesh, Sameer Al Diffalha, David K. Crossman, Changde Cheng, Myng-Hee Kim, Cherlene Hardy, Julienne L. Carstens, Kyoko Kojima, Bart J. Rose, Christopher A. Klug
Abstract
Lung IL-33 is involved in pathogen defense, barrier homeostasis, and development of allergic responses. We previously identified a 5 kb noncoding region within a GWAS-defined segment that regulates expression of human IL33 (hIL33) but is absent in the murine locus. To understand how this region affects IL-33 expression in vivo, we engineered 2 BAC-transgenic strains in which 166 kb of the human genome upstream of the hIL33 locus, along with a fluorescent reporter, was inserted into the murine genome, both with and without the 5 kb region. Comparison to a murine Il33 (mIl33) reporter strain revealed species-specific tropism; hIL33 reporter was mostly expressed in the endothelium, while mIl33 reporter was expressed in type 2 alveolar epithelium. hIL33 reporter expression in tracheal basal epithelium, submucosal glands, and lung microvasculature required the 5 kb region. Surprisingly, allergen and exogenous IL-33 downregulated hIL33 reporter in lung endothelium only when the 5 kb region was present. Similar IL-33–dependent downregulation of IL33 transcripts was observed in human endothelial cell lines, validating that our hIL33 reporter strain recapitulated human endothelial biology. Together, these data reveal the importance of the asthma-associated human 5 kb region in regulating human IL33 expression in a cell type– and context-dependent manner.
Authors
Maile K. Hollinger, Chanie L. Howard, Donna C. Decker, Kelly M. Blaine, Ivy Aneas, Emily M. Grayson, Tania E. Velez, Fernando A. Oliveira, Riley T. Hannan, Daniel F. Camacho, Philip A. Verhoef, Cara L. Hrusch, Rebecca S. Griffes, Jeffrey M. Sturek, Marcelo A. Nobrega, Nathan Schoettler, Anne I. Sperling
Abstract
T cells are the central players in antitumor immunity, and effective tumor killing depends on their ability to infiltrate into the tumor microenvironment (TME) while maintaining normal cytotoxicity. However, late-stage tumors develop immunosuppressive mechanisms that impede T cell movement and induce exhaustion. Investigating T cell migration in human tumors in vivo could provide insights into tumor immune escape, although it remains a challenging task. In this study, we developed ReMiTT, a computational method that leverages spatial transcriptomics data to track T cell migration patterns within tumor tissue. Applying ReMiTT to multiple tumor samples, we identified potential migration trails. On these trails, chemokines that promote T cell trafficking displayed an increasing trend. Additionally, we identified key genes and pathways enriched on these migration trails, including those involved in cytoskeleton rearrangement, leukocyte chemotaxis, cell adhesion, leukocyte migration, and extracellular matrix remodeling. Furthermore, we characterized the phenotypes of T cells along these trails, showing that the migrating T cells are highly proliferative. Our findings introduce an approach for studying T cell migration and interactions within the TME, offering valuable insights into tumor-immune dynamics.
Authors
Lin Zhong, Bo Li, Zhikai Chi, Siyuan Zhang, Qiwei Li, Guanghua Xiao
Abstract
TB (Tuberculosis) and HIV co-infection remains a major global health challenge, with limited understanding of how these pathogens impact local immune responses in the lungs. This study is the first to investigate the modulation of IL-21 during LTBI and Mycobacterium tuberculosis (Mtb)/ Simian Immunodeficiency Virus (SIV) co-infection in non-human primates (NHP). We show that IL-21 expression, predominantly derived from CD4⁺ T cells, is significantly reduced in lungs of Mtb/SIV co-infected macaques, especially in the absence of cART. Although cART and cART with 3HP partially restore IL-21-producing CD4⁺ T cells, levels remain below those in LTBI, indicating ongoing immune impairment. Spatial transcriptomic analysis suggests localized alterations in immune signaling, including differences in STAT1- and STAT3-associated transcriptional profiles and reduced Mtb-specific IFN-γ responses in co-infected animals. Together, our findings indicate that IL-21-producing CD4⁺ T cells are selectively and persistently impaired in the lungs during Mtb/SIV co-infection despite antimicrobial and antiviral therapy. These results highlight a compartment-specific deficit in immune reconstitution and suggest that IL-21-associated pathways may warrant further investigation as potential targets for host-directed therapeutic strategies.
Authors
Vinay Shivanna, Renee D. Escalona, Colin Chuba, Shashi Prakash Singh, Ahmed A. Moustafa, J. Quincy Brown, Chenyao Xiao, Sangkyu Kim, Edward J. Dick Jr., Smriti Mehra, Mirko Paiardini, Riti Sharan
Abstract
BACKGROUND. Chimeric antigen receptor T-cell (CAR-T) therapies have revolutionized treatment for relapsed/refractory multiple myeloma (RRMM). However, cytokine release syndrome (CRS), a common and potentially severe complication, requires inpatient monitoring, limiting access and increasing costs. Wearable devices could support outpatient CAR-T delivery, but feasibility for CRS detection versus standard care remains unproven. METHOD. We conducted a prospective, single-center observational pilot study to assess the feasibility of using wearable devices for monitoring vital signs and detecting CRS. Thirty patients receiving idecabtagene vicleucel (ide-cel) or ciltacabtagene autoleucel (cilta-cel) were enrolled; 25 with sufficient monitoring data were evaluable. Sensors collected skin and axillary temperature, oxygen saturation, respiratory and heart rate, and motion. Peripheral blood cytokines were analyzed pre- and post-infusion using a multiplex proteomic platform. The primary outcome was feasibility, assessed by CRS detection sensitivity and specificity; secondary outcomes included adherence, lead time, and performance of models integrating wearable and cytokine data. RESULTS. CRS occurred in 20 of 25 patients. The best-performing wearable model detected 18 or 20 CRS episodes with a sensitivity of 0.72 (mean 0.75; 95% CI 0.60–0.91) and a specificity of 0.80 (mean 0.76; 95% CI 0.68–0.84), and a median lead time of 7:00 hours before nursing recognition. Median adherence during high-risk periods was 71%. Cytokine changes paralleled temperature elevations, and IFN-γ emerged as a consistent biomarker. CONCLUSION. Wearable devices are feasible for early CRS detection and may support outpatient CAR-T care. Larger outpatient studies are warranted. TRIAL REGISTRATION. This study did not meet the criteria for ClinicalTrials.gov registration.
Authors
Sridevi Rajeeve, Matt Wilkes, Nicole Zahradka, Lewis Tomalin, Mujahid Quidwai, Darren Pan, Nicholas J. Calafat, Martin Cusack, Adolfo Aleman, Kseniya Serebryakova, Katerina Kappes, Hayley Jackson, Sarita Agte, Santiago Thibaud, Larysa Sanchez, Shambavi Richard, Joshua Richter, Cesar Rodriguez, Hearn Jay Cho, Ajai Chari, Sundar Jagannath, Alessandro Laganà, Adriana C. Rossi, Samir Parekh
Abstract
Immune checkpoint inhibitors have transformed cancer therapy, yet many patients fail to achieve durable responses due to insufficient T cell reinvigoration. Cytokines offer promise for enhancing immunotherapy, but their clinical use is limited by toxicity and a narrow therapeutic index. Immunocytokines, engineered fusion proteins combining antibody specificity with cytokine activity, aim to overcome these challenges by targeting cytokine delivery to immune cells or the tumor microenvironment. We describe SAR445877 (SAR’877), a novel PD-1-targeted immunocytokine that fuses a high-affinity anti-PD-1 antibody with a detuned IL-15/IL-15Rα sushi domain complex. SAR’877 blocks PD-1/PD-L1 and PD-1/PD-L2 interactions while selectively delivering IL-15 signals to PD-1+ T cells, enhancing proliferation and activation of antigen-experienced CD8+ and CD4+ T cells and NK cells, while minimizing systemic inflammation. Mechanistically, SAR’877 activates STAT5 signaling in PD-1+ lymphocytes and restores effector function in exhausted T cells. In preclinical models, a murine surrogate of SAR’877 accelerated viral clearance and induced robust anti-tumor immunity by expanding cytotoxic CD8+ T cells and promoting Th1 polarization. Notably, SAR’877 outperformed anti-PD-1 plus untargeted IL-15, highlighting the therapeutic potential of targeted IL-15 delivery. These findings position SAR’877 as a promising next-generation immunotherapy with enhanced efficacy and reduced cytokine-associated toxicities.
Authors
Isaraphorn Pratumchai, Marie Bernardo, Julien Tessier, Jaroslav Zak, Kristi L. Marquardt, Joon Sang Lee, Maheeka Bimal, AHyun Choi, Anthony M. Byers, Mikielia G. Devonish, Roberto Carrio, Dan Lu, Stella A. Martomo, Jeegar Patel, Yu-an Zhang, Ingeborg M. Langohr, Virna Cortez-Retamozo, Dinesh S. Bangari, Angela Hadjipanayis, Xiangming Li, Valeria R. Fantin, Donald R. Shaffer, John R. Teijaro
No posts were found with this tag.