Citation Information: J Clin Invest. 2015;125(1):234-247. https://doi.org/10.1172/JCI77888.
Abstract
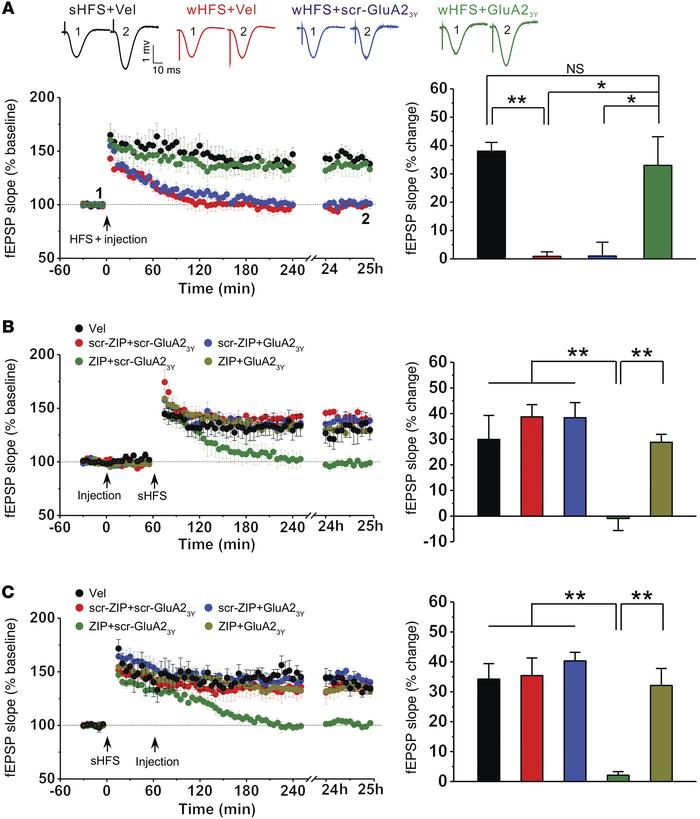
Long-term potentiation (LTP) of synaptic strength between hippocampal neurons is associated with learning and memory, and LTP dysfunction is thought to underlie memory loss. LTP can be temporally and mechanistically classified into decaying (early-phase) LTP and nondecaying (late-phase) LTP. While the nondecaying nature of LTP is thought to depend on protein synthesis and contribute to memory maintenance, little is known about the mechanisms and roles of decaying LTP. Here, we demonstrated that inhibiting endocytosis of postsynaptic α-amino-3-hydroxy-5-methyl-isoxazole-4-propionic acid receptors (AMPARs) prevents LTP decay, thereby converting it into nondecaying LTP. Conversely, restoration of AMPAR endocytosis by inhibiting protein kinase Mζ (PKMζ) converted nondecaying LTP into decaying LTP. Similarly, inhibition of AMPAR endocytosis prolonged memory retention in normal animals and reduced memory loss in a murine model of Alzheimer’s disease. These results strongly suggest that an active process that involves AMPAR endocytosis mediates the decay of LTP and that inhibition of this process can prolong the longevity of LTP as well as memory under both physiological and pathological conditions.
Authors
Zhifang Dong, Huili Han, Hongjie Li, Yanrui Bai, Wei Wang, Man Tu, Yan Peng, Limin Zhou, Wenting He, Xiaobin Wu, Tao Tan, Mingjing Liu, Xiaoyan Wu, Weihui Zhou, Wuyang Jin, Shu Zhang, Todd Charlton Sacktor, Tingyu Li, Weihong Song, Yu Tian Wang
Figure 1



Copyright © 2025 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)