Issue published March 2, 2026 Previous issue

- Volume 136, Issue 5
Go to section:
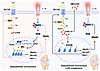
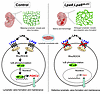
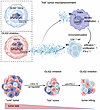
On the cover: Dual role for macrophages in muscle regeneration and degeneration
Yin et al. report that a macrophage subset expressing activin A facilitates muscle regeneration by acting on satellite cells but contributes to heterotopic ossification in pathological settings such as fibrodysplasia ossificans progressiva. The cover image shows injured skeletal muscle sections immunostained with anti–mouse embryonic myosin heavy chain (eMyHC) (magenta), marking regenerating muscle fibers; mouse IgG (light blue), indicating necrotic fibers; and Hoechst (yellow), labeling nuclei. Image credit: Wenqiang Yin, Kazuo Okamoto, and Hiroshi Takayanagi.
In-Press Preview - More
Abstract
BACKGROUND. Estrogen deficiency and progressive hearing loss (HL) are significant concerns in individuals with Turner syndrome (TS). However, whether childhood estrogen deficiency increases HL risk and whether estrogen replacement therapy (ERT) prevents hearing deterioration are still unclear. METHODS. This prospective cohort study recruited children with TS from a tertiary referral center between 2016 and 2024. All participants received standardized recombinant human growth hormone therapy. Longitudinal monitoring data of hormone levels, metabolic parameters, and annual audiological examinations were recorded. The primary analysis used a multivariate Cox model to estimate the adjusted hazard ratio of hearing loss between estrogen-deficient and estrogen-normal TS patients without prior exogenous estrogen exposure. The secondary analysis compared annual pure tone average (PTA) and its changes between the ERT and non-ERT groups in a substudy. RESULTS. Among 87 prepubertal pediatric TS patients, 48 (55.2%) were estrogen-deficient, 38 HL events occurred over 35-month median follow-up. The estrogen-deficient group had higher HL incidence (27 cases, 56.3%; 20.6/100 person-years [PY]) versus estrogen-normal (11 cases, 28.2%; 8.6/100 PY), with estrogen deficiency independently increasing HL risk (HR = 2.93; 95%CI:1.21–7.12). Notably, estrogen deficiency also independently predicted abnormal DPOAE with an even higher effect size (HR = 3.98, 95% CI: 1.35–11.76). The substudy found that initiating ERT at age of 12 significantly preserve auditory function, with the ERT group showing markedly lower PTA and slower hearing deterioration (–1.24 dB/y vs. 1.13 dB/y right ear; –1.85 dB/y vs. 1.04 dB/y left ear, P = 0.001). CONCLUSION. Childhood estrogen deficiency is a modifiable risk factor. Initiating ERT around early adolescence may help hearing preservation. TRIAL REGISTRATION. ChiCTR2300068063. FUNDING. National Natural Science Foundation of China (82173154 and 82471155), Fundamental Research Funds for the Central Universities, Clinical Research 5010 Program, Sun Yat-sen University: 2024004.
Authors
Yan Huang, Liyang Liang, Yanfang Ye, Lina Zhang, Li Ling, Zhe Meng, Wei Liu, Jia Guo, Zulin Liu, Zhen Zhao, Zhigang Zhang, Yu Si
Abstract
Isolating commensal fungi from mouse intestines has been challenging, limiting our understanding of their role in intestinal immune homeostasis and diseases. Using an Fc fusion protein of the C-type lectin Dectin-2, we successfully purified the commensal Ascomycota fungus Engyodontium sp. from mouse feces. Engyodontium enhances the antimicrobial activity of colonic neutrophils via CARD9 pathway, and exacerbates colitis by impairing the colonization of intestinal Lactobacillus johnsonii (L. johnsonii) WXY strain. L. johnsonii produces high levels of L-glutamic acid by expressing the glutaminase-encoding gene glsA to facilitate Treg expansion via enhancing IL-2 receptor signaling. Patients with Crohn’s disease (CD) and ulcerative colitis harbored increased Engyodontium and decreased L. johnsonii abundance. Engyodontium directly induced calprotectin in human colonic neutrophils, and CD patients exhibited lower levels of L-glutamic acid which also promoted human Treg expansion. These findings highlight the Engyodontium-calprotectin axis against the Lactobacillus-glutamate-Treg cascade to aggravate colitis, suggesting commensal Engyodontium-triggered signaling as a therapeutic target for mucosal inflammatory diseases.
Authors
Xinying Wang, Haiyang Sun, Ying Tan, Shaoting Xu, Zishan Liu, Kaile Ji, Ding Qiu, Jianping Deng, Bingbing Feng, Xueting Wu, Yoichiro Iwakura, Minhu Chen, Rui Feng, Chanyan Huang, Ce Tang
Abstract
Osteofibrous dysplasia (OFD) is a skeletal RASopathy presenting with periosteal bone lesions that may progress to fracture and delayed healing (pseudarthrosis). MET gene mutations reducing ubiquitin-mediated protein degradation via loss of the juxtamembrane domain (METΔJMD) were previously identified in OFD patients, resulting in ligand-dependent gain-of-function. The impact of METΔJMD expression on skeletal progenitor cell differentiation and the potential efficacy of targeted therapies remain unclear. We engineered MetΔJMD mice and showed that MetΔJMD expression inhibited osteogenic differentiation of skeletal progenitor cells in vitro and impaired cortical bone development and reduced bone stiffness in vivo. In contrast, conditional deletion of Met enhanced osteogenic differentiation of periosteal progenitor cells. Inhibition of MAPK signaling with MEK inhibitors restored osteogenic differentiation of mouse MetΔJMD skeletal progenitor cells and promoted activation of transcriptional signatures associated with skeletal development and osteoblast differentiation in OFD patient pseudarthrosis-derived primary cells. With this preclinical support, we treated with the MEK inhibitor mirdametinib a pediatric OFD patient suffering from a 3-year history of persistent pseudarthrosis, resulting in fracture union. Our findings demonstrate a bi-directional role for MET in regulating osteogenic differentiation of skeletal progenitor cells and a therapeutic avenue to improve clinical outcomes for this, and potential other, skeletal RASopathies.
Authors
Aysha Khalid, Kristin Denton, Nandina Paria, Ila Oxendine, Meghan Wassell, Reuel Cornelia, Sasidhar Uppuganti, Jeffry S. Nyman, G. Jayashree Jagadeesh, Carlos R. Ferreira, Simon J. Conway, Robert E. Hammer, John O. Ritter, Mylinh Nguyen, David A. Podeszwa, Laura J. Klesse, Carol A. Wise, Jonathan J. Rios
Abstract
Metabolic syndrome and excessive alcohol consumption (MetALD) result in liver injury and fibrosis, which is driven by increased collagen production by activated hepatic stellate cells (HSCs). Our previous studies demonstrated that LARP6, an RNA-binding protein, may facilitate collagen production. However, the expression and function of LARP6 as a regulator of fibrosis development in a disease-relevant model remain poorly understood. We demonstrated that LARP6 was upregulated in human activated HSCs in metabolic dysfunction-associated steatohepatitis (MASH) and MetALD. By using snRNA/ATAC-sequencing, we showed that JUNB upregulated LARP6 expression in activated HSCs. Moreover, LARP6 knockdown in human HSCs suppressed fibrogenic gene expression. By integrating eCLIP analysis and ribosome profiling in HSCs, we showed that LARP6 interacted with mature mRNAs comprising over 300 genes, including RNA structural elements within COL1A1, COL1A2, and COL3A1 to regulate mRNA expression and translation. IP-MS analysis demonstrated LARP6 protein–protein interactions with mRNA translation components and the actin cytoskeleton. Furthermore, dsiRNA-based HSC-specific gene knockdown or pharmacological inhibition of LARP6 attenuated fibrosis development in human MASH and MetALD liver spheroids. Our results suggest LARP6 plays a key role in fibrogenic gene regulation and that targeting LARP6 in human HSCs may represent a therapeutic approach for liver fibrosis.
Authors
Hyun Young Kim, Orel Mizrahi, Wonseok Lee, Sara B. Rosenthal, Cuijuan Han, Brian A. Yee, Steven M. Blue, Jesiel Diaz, Jyotiprakash P. Jonnalagadda, Lena A. Street, Kanani Hokutan, Haeum Jang, Charlene Miciano, Chen-Ting Ma, Andrey A. Bobkov, Eduard Sergienko, Michael R. Jackson, Marko Jovanovic, Branko Stefanovic, Tatiana Kisseleva, Gene W. Yeo, David A. Brenner
Abstract
Neonatal life is marked by rapid antigen exposure, necessitating establishment of peripheral immune tolerance via conversion of naïve CD4+ T cells into regulatory T cells (Tregs). Here, we demonstrated heightened capacity for FOXP3 expression and tolerogenic function among cord blood versus adult blood naive CD4+ T cells and showed that this is linked to their unique metabolic profile and elevated expression of the NADase, CD38. Early-life naïve CD4+ T cells demonstrated a metabolic preference for glycolysis, which directly facilitated their differentiation trajectory. We revealed an age-dependent gradient in CD38 levels on naïve CD4+ T cells and showed that high CD38 expression contributes to both the glycolytic state and tolerogenic potential of neonatal CD4+ T cells, effects that were mediated at least in part via the NAD-dependent deacetylase SIRT1. Thus, the early-life window for peripheral tolerance in humans is critically enabled by the immunometabolic state of the naïve CD4+ compartment.
Authors
Laura R. Dwyer, Andrea M. DeRogatis, Sean Clancy, Victoire Gouirand, Charles Chien, Elizabeth E. Rogers, Scott P. Oltman, Laura L. Jelliffe-Pawlowski, Theo van den Broek, Femke van Wijk, Susan V. Lynch, Rachel L. Rutishauser, Allon Wagner, Alexis J. Combes, Tiffany C. Scharschmidt
View more articles by topic:
Sign up for email alerts
Review Series - More
Series edited by Daniel J. Drucker
Clinical innovation and scientific progress in GLP-1 medicine
Series edited by Daniel J. Drucker
Therapies targeting the glucagon-like peptide 1 (GLP-1) receptor have revolutionized the treatment of obesity and diabetes. This series of reviews, curated by Dr. Dan Drucker, describes the latest research in this fast-moving in field, from our evolving understanding of the mechanism of GLP-1 receptor signaling to the medicines’ impact on inflammation and the consequences for heart, kidney, and brain health. The reviews also explore the impact of these medicines on conditions beyond their initial indications, including cancer and neurodegenerative disease risk.
×